Nitrogen is very unreactive, so high temperatures (500 degrees Celsius) and pressures (200 atmospheres) are required to make it react with hydrogen to form ammonia. The hydrogen gas used for this process is usually obtained from methane derived from natural gas or other fossil fuels. Other authors include graduate students Minju Chung and Kindle Williams, and undergraduate Michal Gala.įor more than 100 years, fertilizer has been manufactured using the Haber-Bosch process, which combines atmospheric nitrogen with hydrogen gas to form ammonia. Graduate student Nikifar Lazouski is the lead author of the paper, which appears today in Nature Catalysis. This could be used by a farmer or a small community of farmers,” says Karthish Manthiram, an assistant professor of chemical engineering at MIT and the senior author of the study.
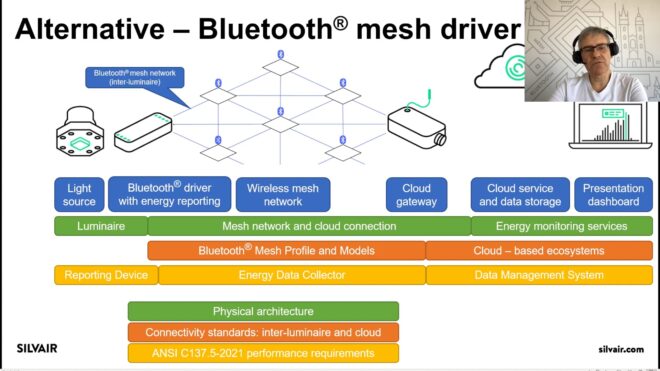
“In the future, if we envision how we want this to be used someday, we want a device that can breathe in air, take in water, have a solar panel hooked up to it, and be able to produce ammonia. In a step toward that kind of small-scale production, the research team has devised a way to combine hydrogen and nitrogen using electric current to generate a lithium catalyst, where the reaction takes place. Fertilizer is often hard to obtain in such areas because of the cost of transporting it from large manufacturing facilities. MIT chemical engineers are working to develop a smaller-scale alternative, which they envision could be used to locally produce fertilizer for farmers in remote, rural areas, such as sub-Saharan Africa. The effectiveness of environmental interventions based on the notion of person-environment fit compared with traditional home hazard checklists remains to be tested.Most of the world’s fertilizer is produced in large manufacturing plants, which require huge amounts of energy to generate the high temperatures and pressures needed to combine nitrogen and hydrogen into ammonia.
The results suggest that much of the inconclusiveness of the data in the relationship between environmental hazards and falls in the previous falls literature could be due to the neglect of person-environment fit assessment. Fallers also experienced lower usability of their home. The person-environment fit problem variable was a stronger fall predictor (odds ratio (OR) = 1.025 P=0.037) than number of environmental barriers (n.s.), even after controlling for confounders.

The number of environmental barriers in the home was similar for the fallers and non-fallers the magnitude of person-environment fit problems was higher among the fallers. The participants reporting falls tended to be frailer than the non-fallers. Urban districts in Sweden, Germany, Latvia.Įight hundred and thirty-four single-living, older adults (75-89 years), in ordinary housing.Īn assessment of objective person-environment fit in the home environment (housing enabler), a self-rating of the perceived home environment (usability in my home) and retrospective self-reports on indoor falls. Survey study with data collection at home visits, followed up by self-reports about falls at home visits one year later. To test the hypotheses that the empirical consideration of objective person-environment fit in the home environment is a stronger predictor of indoor falls among older adults than the assessment of environmental barriers only, and that perceived aspects of home play a role as predictors for falls.
